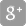Baylis Medical Receives Health Canada Approval for the Ventilators for Canadians COVID-19 Ventilators
The Baylis V4C-560 ventilator is part of the ventilators commissioned by the Government of Canada.
TORONTO, June 16, 2020 /CNW Telbec/ - Baylis Medical and Ventilators for Canadians are pleased to announce today that Health Canada has approved the V4C-560 ventilator. The ventilator is part of the order commissioned by the Government of Canada in support of Canadian patients impacted by COVID-19.
Since the April announcement, Baylis Medical, a developer and manufacturer of cardiology and spine devices, has worked with Ventilators for Canadians, a consortium of Canadian companies, to apply joint advanced development and manufacturing capabilities towards the project.
With the Health Canada approval, manufacturing of the ventilators will now get underway, with the aim of producing the first shipment shortly thereafter.
"We are thrilled to have achieved the first major milestone in our work to serve Canadian COVID-19 patients," said Neil Godara, Vice-President and General Manager of Design and Manufacturing Services at Baylis. "We are grateful for the support of all government agencies involved in this initiative. The ongoing guidance of Health Canada was instrumental in obtaining this approval in record time, while our work with the Public Health Agency of Canada will ensure that the ventilators are deployed without delay. This initiative is truly a team effort."
Rick Jamieson, President and CEO of FTI Professional Grade and a founder of Ventilators for Canadians, added: "The consortium members of Ventilators for Canadians appreciate the collaborative effort, expertise, and commitment from the management teams and employees of Baylis Medical and Medtronic. We also recognize the efforts of the entire supply chain group and the team at Public Services and Procurement Canada, who worked tirelessly to get this project off the ground. Everyone involved knows the significance of helping Canadians during the ongoing COVID-19 pandemic."
The Baylis V4C-560 ventilator, being produced in light of the pandemic, is based on Medtronic's PB560 ventilator design made available through a globally-granted permissive license. The ventilator is compact, lightweight and portable, and provides airway support for both children and adults.
About Baylis Medical
Baylis Medical is a leader in the development and commercialization of innovative medical devices in the fields of cardiology and spine. Headquartered in Canada, and with offices worldwide, our clinical solutions have been Improving the Lives of People Around the World for over 30 years. For more information, visit www.baylismedical.com and connect with us on Twitter, LinkedIn and Instagram.
About Ventilators for Canadians
Ventilators for Canadians (V4C) is a consortium of successful business owners, entrepreneurs, and philanthropists who have come together to identify and pursue solutions to help Canadians through the challenging COVID-19 healthcare emergency. The co-founders of the consortium include; Jim Estill, CEO of Danby and ShipperBee, Rick Jamieson, President and CEO of FTI Professional Grade and ABS Friction, Paul L'Heureux, CEO of Crystal Fountains, and Scott Shawyer, President & CEO of JMP Solutions.
PRM-00609 EN J-1,2,3 V-1 © Baylis Medical Company Inc., 2020. The Baylis Medical logo is a trademark and/or registered trademarks of Baylis Medical Company Inc. in the USA and/or other countries. Other trademarks are the property of their respective owners.
SOURCE Baylis Medical Company Inc.


Upcoming Life Sciences Events
- April 2024
- London: BioTrinity 2024
- Singapore: Asia Bio Partnering Forum
- London: LSX World Congress
 Biotechgate Global Database ›
Biotechgate Global Database ›